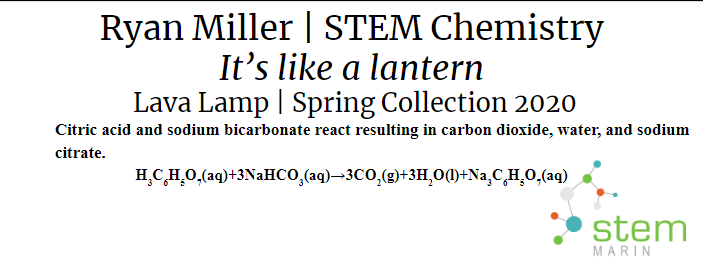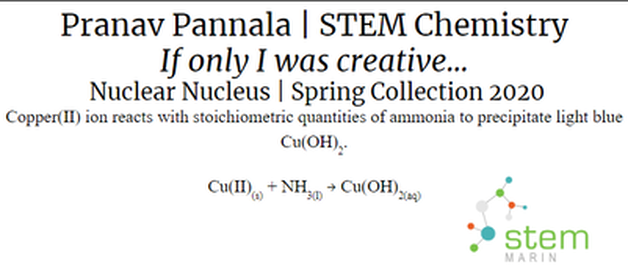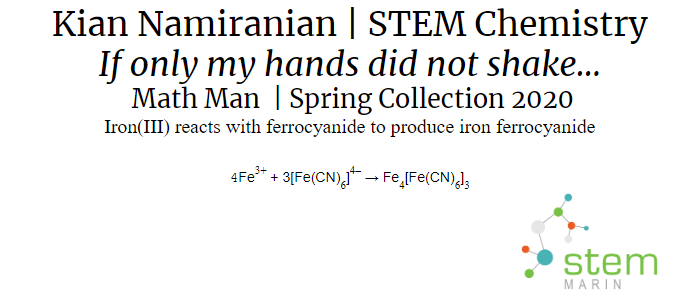Evidence of Work
In this project our groups were tasked with each creating four art pieces by using chemistry that we had learned. My group decided to each make one art piece following a glow in the dark theme. We also each incorporated one of the four letters of stem into our art pieces.
Lava Lamp |
Nuclear Nucleus |
|
I made a Lava Lamp by putting water and vegetable oil into a clear container. The vegetable oil floats to the top because it is less dense. A light is also placed underneath so that the Lamp glows while in darkness. Alka Seltzer tablets are then dropped into the Lamp which causes an Acid-Base reaction when mixed with water resulting in Carbon Dioxide which creates a bubbling affect in the vegetable oil.
|
In his project, Pranav created a model of an atom by connecting pieces of copper and making a piece of art. He then coated it with a glow in the dark paint so that it would glow in the dark and fit with the theme.
|
Epic Engineering |
Math Man |
|
In his project, Evan used a 3D printer with glow in the dark filament to make an art piece. The model was designed using a program called Tinkercad and then printed with his 3D printer. It has gears on the inside allowing it to spin by cranking a lever. The glow in the dark filament allows it to absorb light and then glow in the dark when in darkness.
|
For his project, Kian made a painting using Prussian Blue paint that he created as well as glow in the dark paint to make a glow in the dark painting of the mathematician George cantor. The Prussian Blue was a participate that was created by a chemical reaction between Iron and Ferromagnetic.
|
Content
|
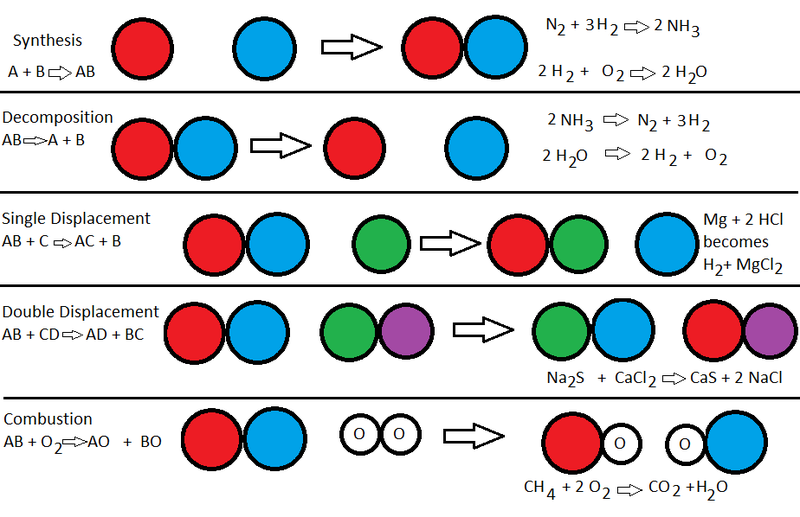
Types of Reactions: There are 5 main types of reactions we learned about, Synthesis, Double Displacement, Single replacement, Decomposition, and Combustion Reactions. In a synthesis reaction two reactions are combined to form a single product. In a Double Displacement Reaction two reactants trade components to create two products. In my lava Lamp I used a double displacement reaction, specifically an acid-base reaction to create carbon dioxide. A single replacement swaps one component from a reactant with an ion to create a product. In a decomposition reaction a single reactant becomes two products. In a combustion reaction needs heat to occur and results in water and carbon dioxide.
|
|
Balancing Chemical Reactions: We learned how to balance chemical equations. In order to balance a chemical equation one must make sure there is an equal number of each specific element on each side. It is also important to make sure that all the compounds are neutral. If you have 6 Hydrogen on one side, for example, it is important to keep track of the amount of hydrogen on the other side and make it six in order to balance the equation.
Chemical Formulas and Names: During this project we also learned how to go from the written form of an equations to the chemical equation and vice versa. There are many general trends that one can follow to allow us to easily go from words to symbols. We used this knowledge in our project in order to make sure we had the right chemicals for our reactions. Reactivity series: In order to find out whether a single replacement reaction will occur it is important to understand the reactivity series. It is a list of compounds in an order. If the compound is higher on the list then it can trade out with the compound lower on the list inside the reactant. |
Reflection
In this project the two things I did well were having a good work ethic and critical thinking. I was able to finish all of my work successfully and create my presentation on time. I also was able to think creatively and create an art project that looks okay in the dark. Two things that I need to work on for the next project are collaborations and leadership. I failed to successfully collaborate with my teammates to the effectiveness that I could. I feel that it was due to the nature of the project being that we each separately created art pieces. This also led me to not have good leadership as I was not really collaborating or leading the rest of my group in this project. In future projects I will try to collaborate with my teammates more and work as a group.